In the traditional off-line quality control, samples are taken at various process stages and transported to a quality lab for analysis. It may take hours until the results are available, resulting in delays that make results-based process control impossible. In a PAT-based approach, the pharmaceutical manufacturing process and the analytical instruments, such as spectrometers, acquire in-line measurement results in real time. This real-time data can improve both process control and product quality.
Key facts
> 1500
Raman spectroscopy units installed worldwide
Increase process control and product quality with PAT

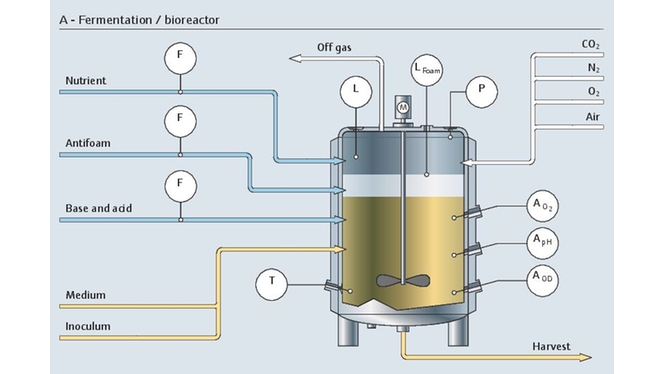
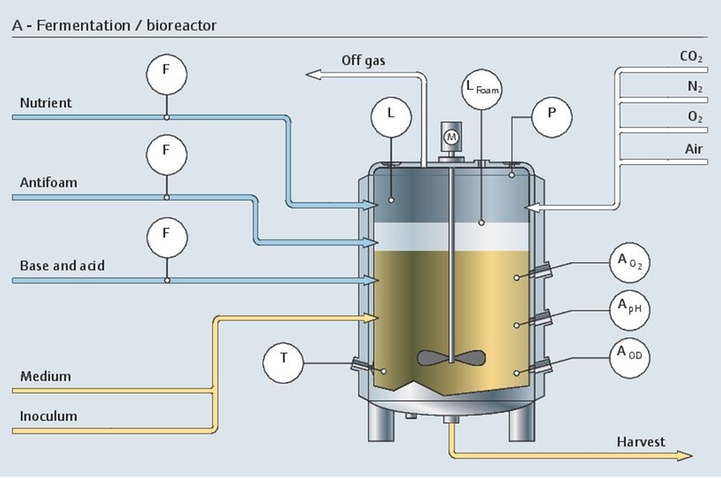
Optimized process control in bioreactors
In a bioreactor, it is important to keep the dissolved oxygen and pH at an optimal level at all times. Cell density and metabolites should also be monitored for a better insight into the process. Metabolite concentrations are typically measured off-line with time-consuming techniques and therefore require sampling with the associated risks.
Our expertise in the field
Raman spectroscopy is an ideal solution for fast and non-destructive measurement. Our Raman solutions can monitor several parameters simultaneously with one single probe installed directly in-line. The complete upstream package enables real-time and optimized control of the process - leading to reduced time between batches, limited risk of contamination and increased yield.

In-line glucose control
Glucose is the main energy source of cells during the upstream process. The concentration of glucose is a critical process parameter (CPP) because excessive glucose can lead to metabolic inefficiency and subsequent toxic lactate and glucose accumulation. Traditionally, a sample must be taken from the cultivation and analyzed in the lab to determine the amount of glucose that needs to be supplied to the bioreactor.
Our expertise in the field
Direct in-line monitoring of glucose concentration provides a better understanding and control of the fermentation or cell culture process. Continuous glucose measurements using Raman analyzers not only offer important bioprocess information for better process understanding but also bring huge potential for optimization. For example, Raman-based glucose control in a mammalian cell bioprocess has enabled titer increases of up to 85%.
- Raman spectroscopy technology is easily integrated into bioreactor controllers, and is a proven PAT solution able to provide real-time process control and quality assurance

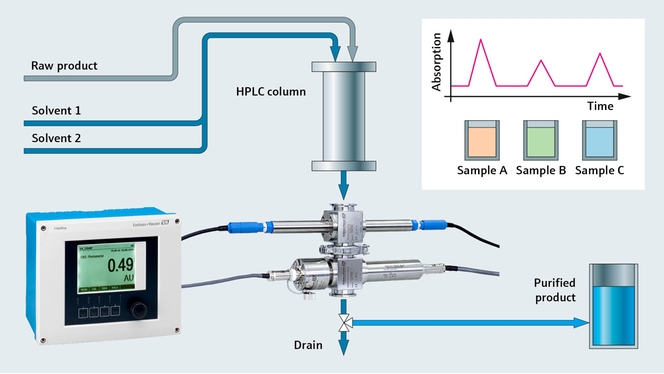
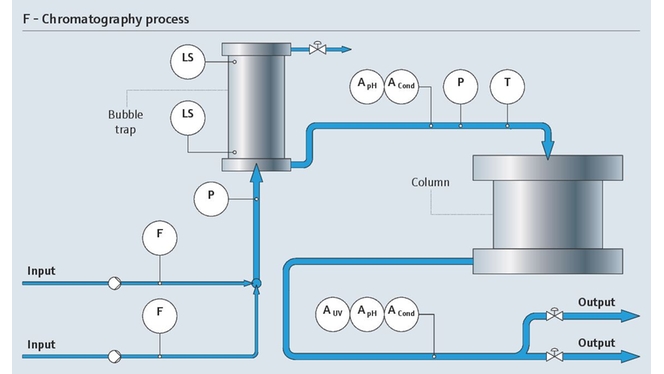
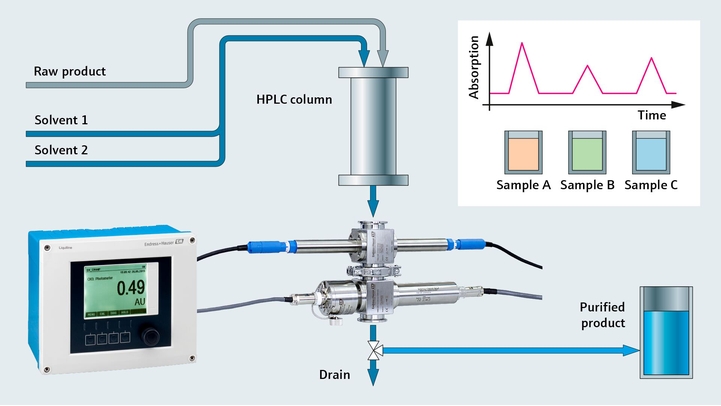
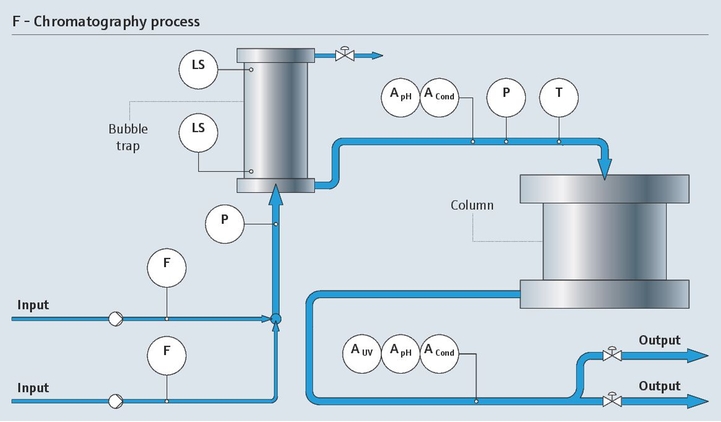
Protein monitoring in chromatography
As a pharmaceutical manufacturer, you strive for highest possible yield and purest product in your downstream process. Improving chromatography and purification of your product mixture is a key aspect in achieving this goal. To reach maximum purification with minimal loss, precise and sensitive UV absorption with fast and reliable protein detection is required.
Our expertise in the field
With our UV absorption photometer, we bring the sensor and the measurement directly into the process by measuring protein concentration in-line. For signal processing we use our well-known transmitter platform, Liquiline. Using only a single Liquiline CM44P transmitter, you can also add pH and conductivity sensors, making it an ideal solution for chromatography control.
- The OUSAF44 UV sensor monitors the concentration of your product in process liquids and is suitable for sterile processes (CIP/SIP)
- The low-volume flow cell OUA260 for the process photometers enables fast measurement and ensures optimum product yield. For pH and conductivity, the flow cell CYA680 is the right choice
- The fully encapsulated Easycal system provides safe, liquid-free online calibration of UV sensors, traceable to NIST
Benefits
With inline spectrometers, measurement frequencies in the range of seconds can be achieved, thus enabling real-time advanced process control (APC) in pharmaceutical manufacturing. Adopting process analytical technology (PAT) helps develop processes that produce higher-quality products and improve efficiency through increased automation, reduced cycle times and uninterrupted batch or continuous processing.
Key facts
35
fermentation batches until ROI of a single-channel Raman Rxn system
Key facts
10x
lower maintenance costs of a Raman measurement compared to a chromatography system
Key facts
90%
less time spent calibrating one UV sensor with Easycal
How we can help
We help you achieve advanced process control through in-line measurement of critical quality parameters. Analysis results are available in seconds instead of hours - shifting from time-consuming sample analysis to real-time monitoring of critical process parameters or critical quality attributes. With our optical technologies, such as Raman and UV spectroscopy, you can monitor and control the concentration or composition of your process media or final product in real time. Your benefits are:
- improved process control leading to optimized production, increased yield and higher product quality
- elimination of contamination risks and time savings through in-line process monitoring without the need for sampling
- complete packaged solutions for upstream and downstream processes
- simplified scale-up with probes suitable for all scales and technologies (e.g., glass, stainless steel, single-use plastic)